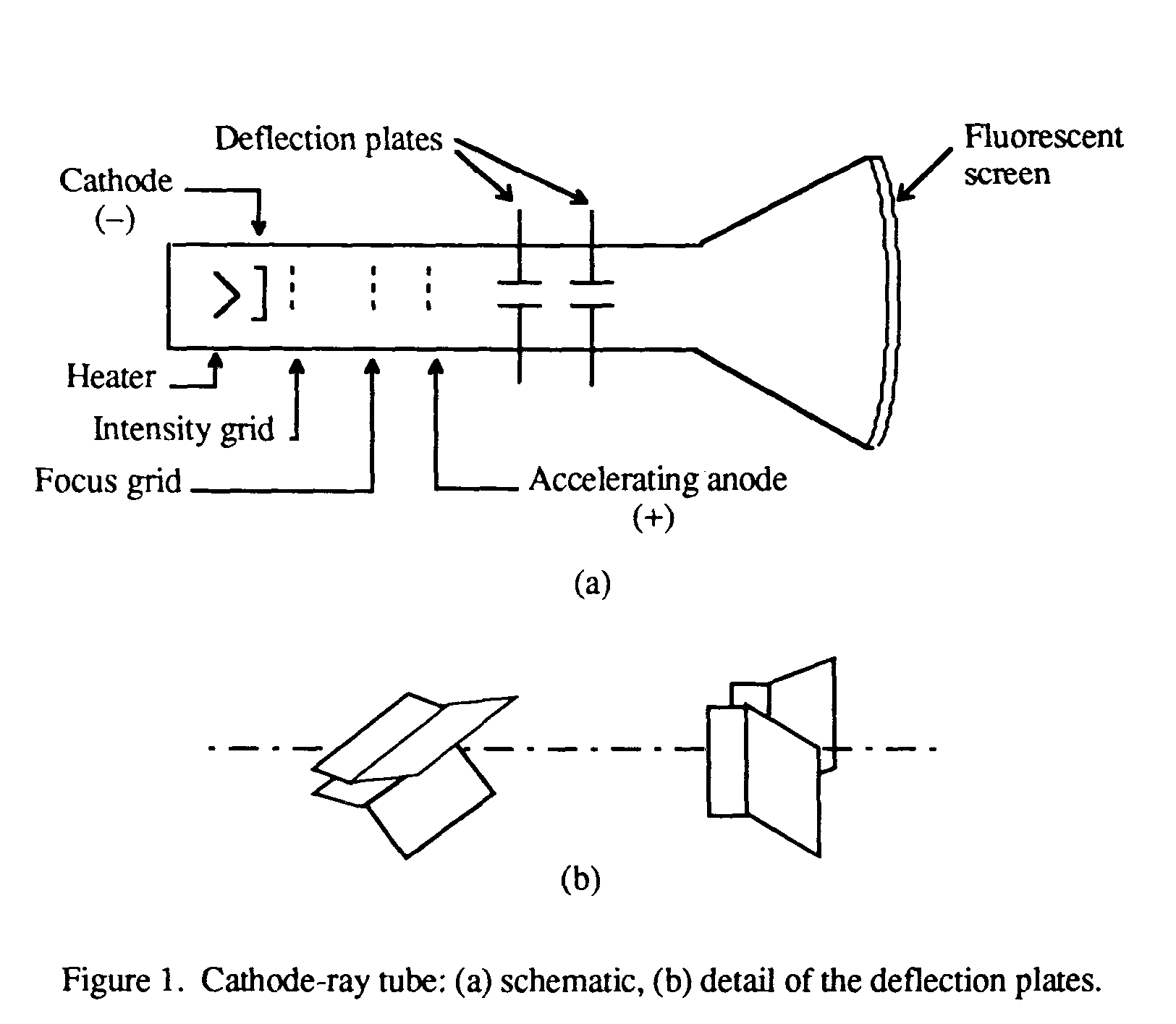
These cylinders were closed except for two small holes, one in each cylinder, placed so that the cathode rays could pass through them into the inside of the inner cylinder. This has been proved to be the case by Perrin, who placed in front of a plane cathode two coaxial metallic cylinders which were insulated from each other: the outer of these cylinders was connected with the earth, the inner with a gold-leaf electroscope. If these rays are negatively electrified particles, then when they enter an enclosure they ought to carry into it a charge of negative electricity. The following experiments were made to test some of the consequences of the electrified-particle theory. The electrified-particle theory has for purposes of research a great advantage over the aetherial theory, since it is definite and its consequences can be predicted with the aetherial theory it is impossible to predict what will happen under any given circumstances, as on this theory we are dealing with hitherto unobserved phenomena in the aether, of whose laws we are ignorant. It would seem at first sight that it ought not to be difficult to discriminate between views so different, yet experience shows that this is not the case, as amongst the physicists who have most deeply studied the subject can be found supporters of either theory. The most diverse opinions are held as to these rays according to the almost unanimous opinion of German physicists they are due to some process in the aether to which-inasmuch as in a uniform magnetic field their course is circular and not rectilinear-no phenomenon hitherto observed is analogous: another view of these rays is that, so far from being wholly aetherial, they are in fact wholly material, and that they mark the paths of particles of matter charged with negative electricity. 
The experiments * discussed in this paper were undertaken in the hope of gaining some information as to the nature of the Cathode Rays. These subatomic particles can be found within atoms of all elements.Ĭonclusion - The cathode rays are made up of negatively charged particles known as electrons.J. Thomson also placed two magnets on either side of the tube, and observed that this magnetic field also deflected the cathode ray. 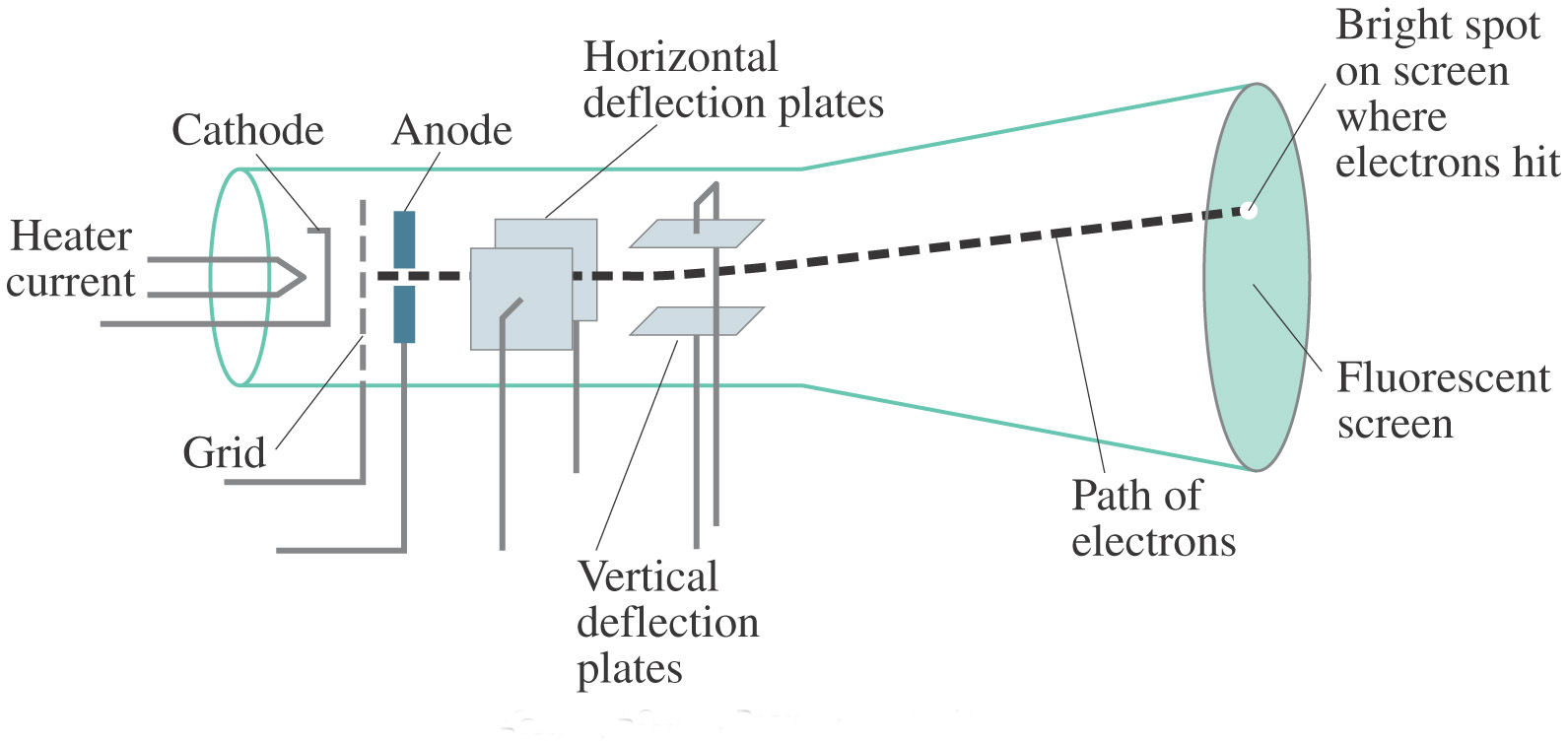
This indicated that the cathode ray was composed of negatively-charged particles. The cathode ray was deflected away from the negatively-charged electric plate and towards the positively-charged plate. Thomson's experiment on constituents of cathode rays:Įxperiment - To test the properties of the particles, Thomson placed two oppositely-charged electric plates around the cathode ray. An electric discharge is passed through the tube.Įmission of blue rays can be seen from the cathode.Ĭonclusion - The blue rays emitted from cathode are cathode rays. William Crookes experiment for the discovery of cathode rays:Įxperiment - The figure shows a vacuum tube which has a gas at a very low pressure.The discovery of cathode rays lead to the discovery of electrons : The three subatomic particles that constitute an atom are:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
